 NIST Chemistry WebBook, NIST Standard Reference Database No. 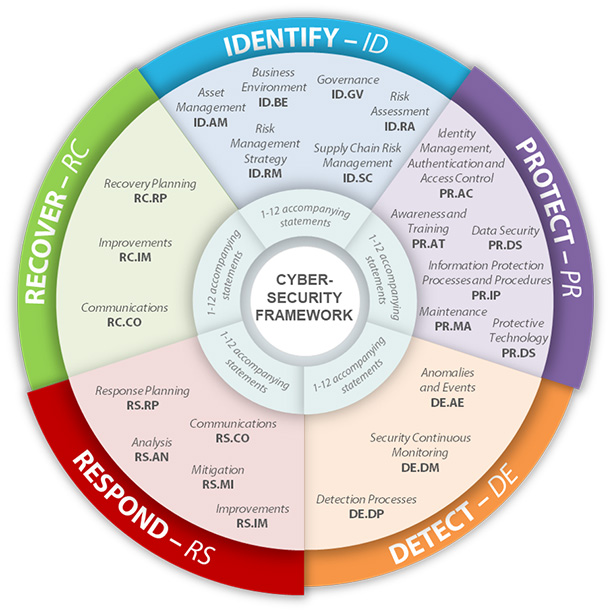
The BrHF geometry was optimized using MP2(Full) /6-311+ G (3df ), then the G2 total energy was calculated for this geometry. Linstrom (National Institute of Standards and Technology, Gaithersburg, MD, 1998) (). Lias, Proton Affinity Evaluation (WebBook), in NIST Standard Reference Database Number 69, edited by W. Schlegel et al., GAUSSIAN 98, Gaussian, Inc., Pittsburgh, PA, 1998. These new measurements bring into harmony AE ( CF 3 + / CF 4 ) obtained from the HCl + reaction and the value AE ( CF 3 + / CF 4 )=14.67☐.04 eV reported by R. National Institute of Standards and Technology. The experimental data shown in these pages are freely available and have been published already in the DDB Explorer Edition.The data represent a small sub list of all available data in the Dortmund Data Bank.For more data or any further information please search the DDB or contact DDBST. The rate constant for thermal decomposition of HCF 4 + into HF + CF 3 + was measured in the course of this work. For information about purchasing and accessing your subscription: Standard Reference Data Program. In the present work, the rate constant was found to be collisional, producing HCF 4 +. This standard reference material from NIST will help calibrate procedures for calcium determinations used in clinical analysis and for routine critical. 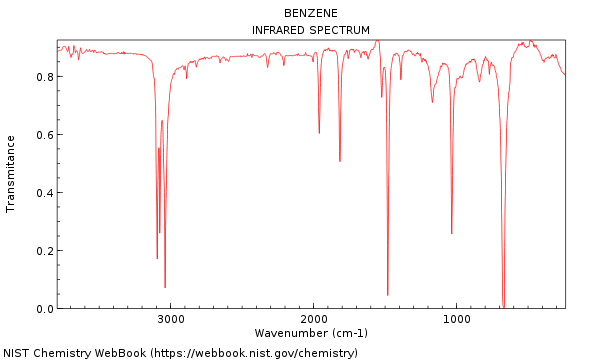
A remeasurement of this reaction over the temperature range 173–500 K shows that the original study was in error, both in the reported rate constant and the ion product. SRM 915c Calcium Carbonate Mass Fraction Standard. In an earlier work the reaction between HCl + was used to support a low value, AE ( CF 3 + / CF 4 )=14.2 eV. This is indicated by underling and highlighting the species. Presently this includes direct links to the corresponding NIST WebBook page for all substances for which such a link is possible. toxic substance data : information provided by: .gov (external link) Last Update 18:28:29. produced from DL-methionine as a raw material. or with sodium cyanate and hydrochloric acid to produce meprobamate. 
For some years there has been a dispute concerning the appearance energy of CF 3 + from CF 4, or alternatively, the ionization energy of CF 3 or the heat of formation of CF 3 +. The Kinetics Database is being expanded to include other resources for the convenience of the users. information provided by: (external link) EPA chemical substance information. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
